
CSI HIGHLIGHT OF THE YEAR award:
Congenital
CSI Awards celebrate highlights of the year, events such as first-in-mans, trials, studies and approvals, among others, within the field of congenital, structural and valvar heart disease interventions and interventional imaging.

Proven PDA Closure for patients 700g and up. Closes Early PDAs. Fills Loving Hearts
Abbott | congenital
Impact outline: The Amplatzer ADOIIAS was originally developed as a device to close PDAs in premature infants. Unfortunately, the device was never approved for the originally intended indication. In the early days, this was not a real problem.Today, however, off-label use is not as well accepted. Although it does not seem to affect the senior implanters, it restricts regulatory approvals and alliance to use in certain countries. Moreover, it prevents promoting devices to referrals, such as neonatologists.
Background/supporting information: To get this device on label for premature infants, Abbott initiated the ADOIIAS – IDE trial. With collaboration and guidance from the FDA this 50-patient study was designed. After completing the study, Abbott renamed the device Amplatzer Piccolo™ Occluder. The Amplatzer Piccolo demonstrated safety and effectiveness with a low rate of major complications and a high rate of PDA closure. It received FDA approval in record time on January 15th.
As the only PDA closure solution indicated for premature infants (≥700g + ≥3 days old) and proven to deliver safe and effective closure, Amplatzer Piccolo Occluder offers new opportunities to care for a wider range of patients than ever before. Abbott is committed to developing minimally invasive life-saving pediatric devices that have an immediate impact with long-term benefits, reduce the risks of life-threatening complications and allow physicians to confidently treat the youngest and tiniest patients.
As a for-profit company, products like the Amplatzer Piccolo, in comparison to other structural heart device opportunities, do not have a huge sales and the market opportunity.. Still, we feel that this patient population is important and that we need to contribute to the medical technology field with these new, life changing innovations.
* AMPLATZER PICCOLO™ OCCLUDER pending CE mark. Not available for sales outside the USA.
* * *
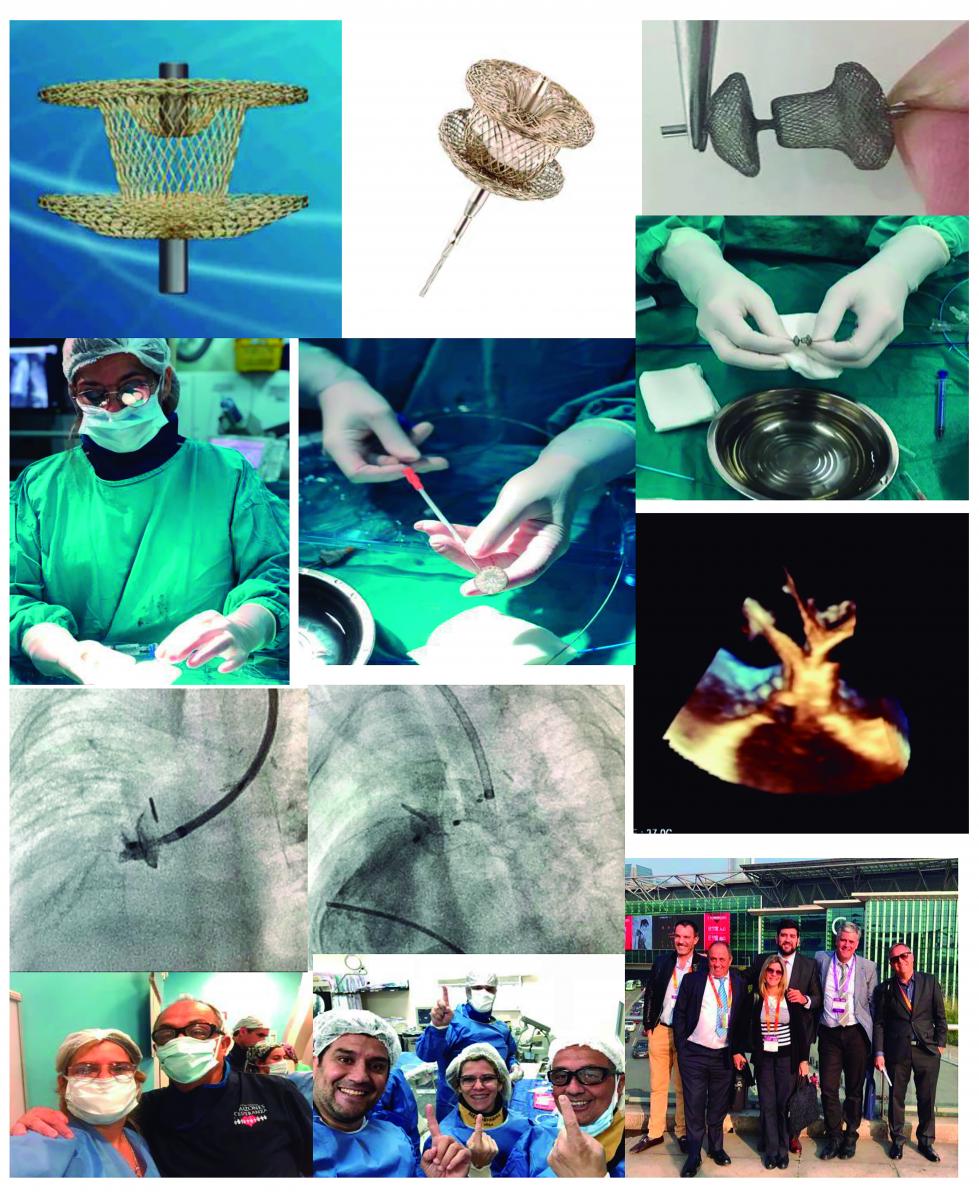
Konar multifuncional occluder: A device to treat different heart diseases
lifetech | CONGENITAL
Impact outline: The Konar-MF multifunctional occluder(MFO) is a new soft woven mesh self-expanding device with 2 discs joined by an articulated waist. Each discs has a hub on the external surface, so the device can be either retrogradely or anterogradely positioned. It is a sleek device and delivery cable
Device closure is now an accepted modality of treatment for cardiac septal defects, patent ductus and delivery cablecardiac septal and non septal defects, patent ductus arteriosus and other selected defects to treat successfully with the Konar-MF device occluder without significant complication and with more options in terms of approach of each one of them. The low profile in different measures allows closure of large defects even in low weight patients.
This device will let us to treat a varied number of congenital heart deseases as VSD, PDA, any kind of Fistula, RVOT, MAPCAs and any other that it conform to specific morphology.
Background/supporting information: Percutaneous ventricular septal defect (VSD) closure remains a complex intervention [1]. In the patient population requiring this intervention, small, flexible and low-profile devices may offer good closure rates with a low rate of complications [1, 2]. Previously, off-label use of patent ductus arteriosus (PDA) devices (e.g. Amplatzer PDA devices; ADO I and II) has been reported in several case series [1-3]. In May 2018, the KONAR-MF (MF= Multi Functional) TM VSD device received CE-mark approval for VSD closure after first in man implantation in 2013.
First European experience of percutaneous closure of ventricular septal defects using a new CE-marked VSD occluder was done from November 2017 to april 2018 in fiur patients. We have reported the first experience in Latin America from October 2017 to November 2018 of VSD closure with the KONAR MFO device (in press).
* * *
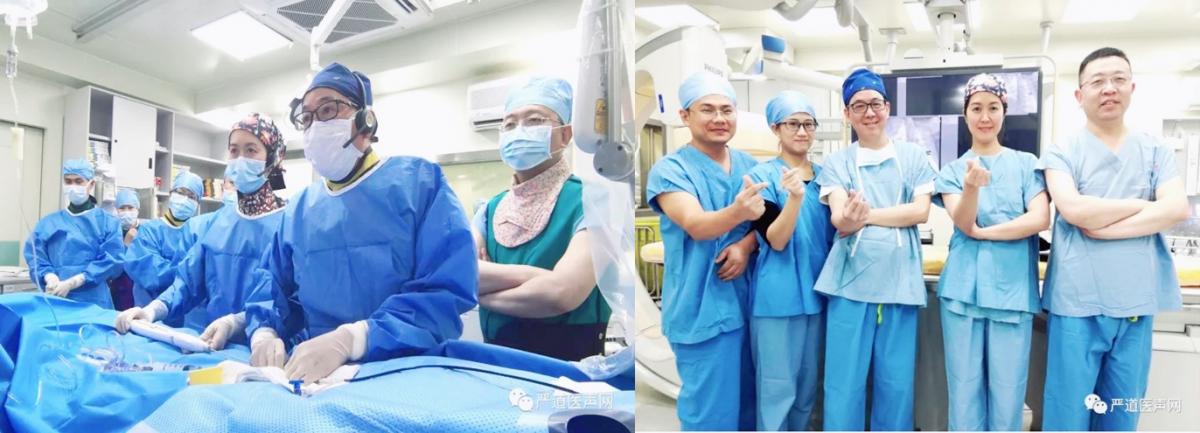
CSI AP 2019 Prof. Zhiwei Zhang ’s team performed global live case transmission with Absnow absorbable ASD Closure System for the congenital atrial septal defect
LIFETECH | CONGENITAL
Impact outline: On February 23, 2019, during CSI AP 2019,professor Zhang Zhiwei's team from Guangdong provincial people's hospital of China implanted an absorbable atrial septal closure device which independently developed by China for a 5-year-old child with congenital atrial septal defect who suffered repeated pneumonia.
Background/supporting information: Why Absorbable occlude? Metal frameworks may cause erosion, arrhythmia, thrombus formation, and aortic valve leaflet deformity. To make future trans-septal access possible, such as EP procedure, mitral valve intervention and LAA closure. No residue in the body, patients prefer it.
* * *
Bifurcating Stents in the Pulmonary Arteries: a Specially Designed Stent
Manufactured in China for the Treatment of Branch
Pulmonary Artery Stenosis
med zanith | CONGENITAL
Impact outline: Stent treatment in close proximity to the bifurcation of the branch pulmonary arteries is a challenging problem. We successfully used a specially designed stent manufactured in China to treat bilateral branch pulmonary artery stenosis. The procedure involved dilating a cell of the RPA stent, simultaneous RPA and LPA dilations and stent placement. Angiogram after intervention demonstrated relief of bilateral pulmonary arteries obstructions.
Background/supporting information: Since O 'Laughlin, et al. first reported stent implantation to treat pulmonary artery stenosis, the procedure has rapidly gained acceptance, but all of these stents were “off label” and not designed for this purpose. Pul-Stent™ was specially designed for pulmonary artery stenosis. It is composed of the newer and stronger cobalt alloy, rather than stainless steel. It has both of the open-cell and closed-cell stent advantages: ability to cross side branches and be redilated and minimal shortening, as well as a higher radial strength and a low profile.
* * *

A self-expanding percutaneous pulmonary valve specifically for the native RVOT
Venus medtech | CONGENITAL
Impact outline: Percutaneous pulmonary valve implantation was initially limited to patients who had circumferential surgical right ventricular outflow tracts (RVOT), for example conduits or bioprosthetic valves, but its efficacy is now being demonstrated in patients who have had RVOT reconstructions with a transannular patch. RVOT size remains a critical determinant of suitability for PPVI.
The Venus P Valve is a new percutaneous pulmonary valve designed specifically for the native RVOT. Its self expanding design can be accommodated in RVOT diameters up to 32 33mm which is larger and more curved structure of the RVOT than commercially available valves. Furthermore, there is no need for pre stenting to create a landing zone. Clinical experience has been encouraging suggesting that the Venus P Valve can be implanted safely with good short and medium term results in patients with native right ventricular outflow tracts.
Background/supporting information: The Venus P Valve consist s of a stent of a Nitinol frame, and the valve leaflets are made of porcine pericardium preserved in low concentration solutions of buffered glutaraldehyde and are hand sewn to the from 18 36mm (with 2 mm increments) and from 20 35 mm (with 5 mm increment), respectively. The proximal and distal flares generate an hourglass configuration which help to stabilize the valve in the RVOT and main pulmonary artery. The delivery systems consists of a 20 22 Fr capsule and a 16 Fr 100 cm long shaft .
The Venus P Valve has been developed to address the challenges posed by the dilated native outflow tract, making it possible to implant valves in a wider range of patients, especially those patients who previously had a surgical transannular patch repair of tetralogy of Fallot.
* * *

Bioabsorbable Iron stent for PDA
Lifetech | CONGENITAL
Impact outline: PDA stenting using bioresorbable Iron stent in a neonate with duct-dependent lesion - Why biodegrabable stents are desirable in infants and small children:
1. Avoid limiting vessel diameter associated with bare metal stent (BMS); largest coronary BMS is 4.5 mm; stents dilatable to adult size vessels - too bulky for implantation in infants.
2. Once stent degrades – allow further stent implantation, use of cutting balloon etc.
3. Potential application – Pulmonary artery stenosis, pulmonary vein stenosis, RVOT in Fallot Coarctation Coronary.
Background/supporting information: Preliminary results of IBS stents in PDA with simple morphology: acute and early post procedure results of IBS stent for initial palliation of cyanotic CHD with duct dependent pulmonary is a promising. However this pilot study is limited to patients with simple ductus morphology (PAIVS and critical PS patients).
1. Ease of implant and x-ray visibility similar to BMS, pulmonary overcirculation seems a common problem.
2. 3 mm stent instead of 4 mm in patients with dual source PBF.
3. Need for dual antiplatelet once overshunting settles.
* * *